Does #"H"_4"O"_2^(+)# exist in water?
1 Answer
Oct 21, 2017
No, because the charge is not conserved...
#"H"_5"O"_2^(+)# (i.e. the Zundel cation)#"H"_7"O"_3^(+)# #"H"_9"O"_4^(+)# (i.e. the Eigen cation)
etc.
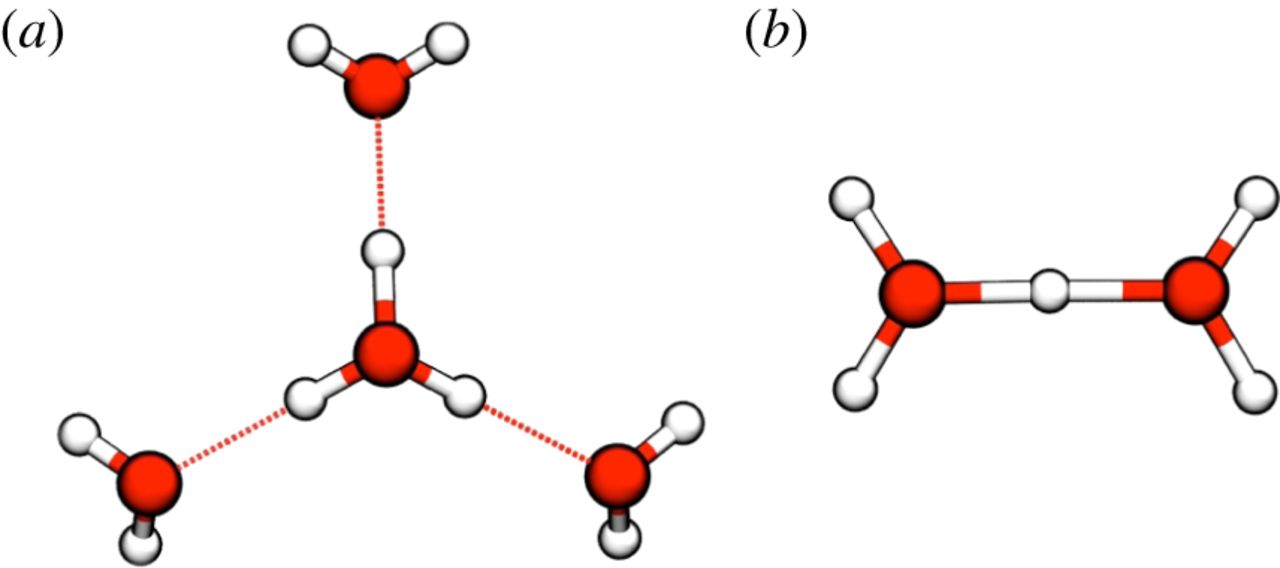
But we only need to speak of

